tooth whitening
‘the act or process of making or becoming white’
teeth whitening involves bleaching your teeth to make them lighter. the chemical degradation of the chromogens within or on the tooth is termed as bleaching. it can’t make your teeth brilliant white, but it is a very effective way of lightening the existing colour by several shades without removing any of the tooth surface. once the teeth are whitened, it is common to notice a facial radiance similar to having a suntan
this in turn creates a huge ‘feel good’ factor and boosts confidence.
how does it work? – the science!
 hydrogen peroxide is the active ingredient most commonly used in whitening products and is delivered as either hydrogen peroxide or carbamide peroxide.
hydrogen peroxide is the active ingredient most commonly used in whitening products and is delivered as either hydrogen peroxide or carbamide peroxide.
hydrogen peroxide is comparable to carbamide peroxide as it is released when the stable complex is in contact with water.
as it diffuses into the tooth, it acts as an oxidising agent that breaks down to produce unstable free radicals.
in the spaces between the inorganic salts in tooth enamel, these unstable free radicals attach to organic pigment molecules resulting in small, less heavily pigmented components.
these smaller molecules reflect less light and hence create a “whitening effect”.
time exposure and the concentration of the bleaching compound, determines the tooth whitening endpoint.
types of whitening treatments
custom made home bleaching trays

impressions are taken of the upper and lower teeth.
custom trays are then made allowing for placement of gel within.
a peroxide gel is placed in the trays and inserted into the mouth and worn overnight.
the gel is most active for the first 2 hours. the efficiency then decreases by around 50%.
a shade difference is usually seen after 3 nights of wearing the trays.
fantastic because it is controllable (keep going until you’re happy with the result),
reusable (keep the trays safe and reuse them in a year or two) and generally cheaper.
in surgery – laser/power whitening
bleaching procedures generally use a light-cured protective layer that is carefully painted on the gums and papilla (the tips of the gums between the teeth) to reduce the risk of chemical burns to the soft tissues.
the bleaching agent is either carbamide peroxide – which breaks down in the mouth to form hydrogen peroxide, or hydrogen peroxide itself.
the gel typically contains between 10% and 44% carbamide peroxide, which is roughly equivalent to a 3% to 16% hydrogen peroxide concentration.
power or light-accelerated bleaching uses light energy which is intended to accelerate the process of bleaching.
different types of energy can be used in this procedure, with the most common being halogen, led, or plasma arc.
internal bleaching
often sought after in teeth which have been endodontically treated as tooth discolouration becomes a problem due to the lack of nerve supply.
a way around this is by sealing off the bleaching agent inside the tooth itself and replacing it every few weeks until the desired shade has been achieved.
it is common to have this done on a front tooth for aesthetic reasons.
over the counter
there are many ‘home’ whitening tools on the market. from toothpaste, rinses and strips to entire kits.
it is big business.
however, to be deemed safe for home use, the quantity of active ingredient is small, therefore the effect will usually be very, very gradual sometimes taking years.
it is worth noting:
the legal percentage of hydrogen peroxide allowed to be given is 0.1–6%.
bleaching agents are only allowed to be given by dental practitioners, dental therapists, and dental hygienists.
bleaching is most effective with yellow discoloured teeth.
it is least effective when the original tooth colour is greyish.
if heavy staining or tetracycline damage is present on a patient’s teeth, and whitening is ineffective (tetracycline staining may require prolonged bleaching, as it takes longer for the bleach to reach the dentine layer), there are other methods of masking the stain.
eg. bonding/veneers
tooth straightening
‘make tidy or put in order again’
the branch of dentistry called orthodontics.
there is nothing quite as nice as the perfect smile.
it is attractive, welcoming and inviting.
no surprise then, that over the years, parents worldwide have encouraged their children to undergo orthodontic treatments in order to correct any ‘crookedness’ of the dentition!
as mankind has evolved our diets have changed – less raw, more processed – resulting in a gradual shrinking of our jaws.
commonly this in turn means there is not enough room for all our teeth.
no doubt as we evolve more, we will, in time, also have less teeth…. evolution will sort it!
hence, orthodontics.
it is very complicated but in essence involves moving teeth slowly through the jaw bone so as not to cause damage/death to the bone, teeth or surrounding tissues.
the aim is to achieve the best alignment possible.
there are many, many different types of retainers (more commonly called ‘braces’) available.
fixed, removable, lingual etc.
typically treatment should be sort from a specialist orthodontist.
Invisalign®

previously straightening teeth was for kids.
adults generally did not want to wear the ‘train-tracks’ as they were unsightly. not anymore! luckily, the advent of computer aided design and technology has now opened up a whole world of clear removable aligners which can move teeth from ‘a to b’ with minimal intervention, achieving maximum effect. each aligner is worn for 22hrs a day. 7 days a week. the results are amazing
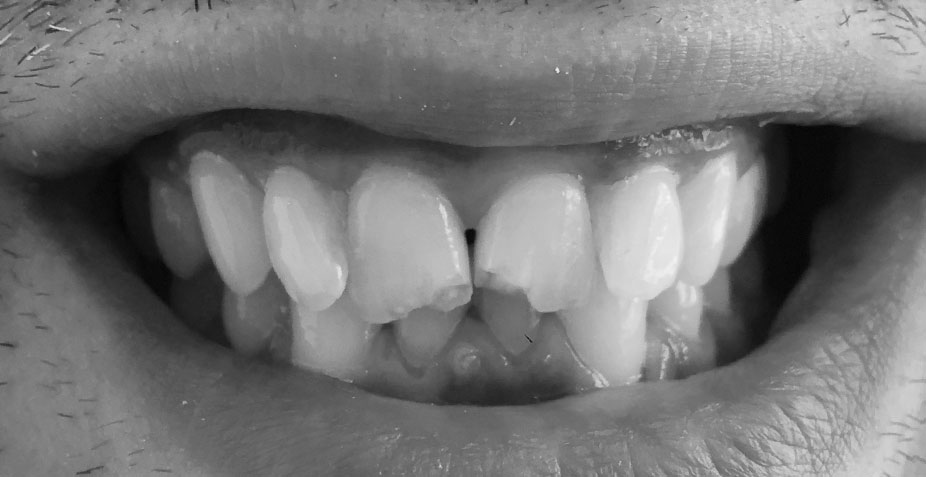
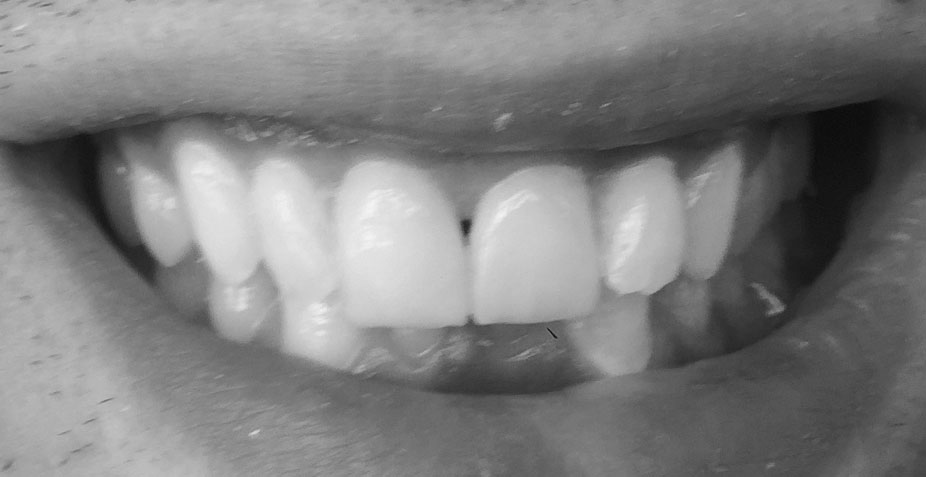
tooth veneers
‘to overlay with a thin layer of fine or decorative material’
tooth veneers are wafer-thin, custom-made shells of tooth-colored material designed to cover the front surface of teeth. basically false fingernails for teeth. clever, in that they can be used to close gaps, appear to straighten teeth or permanently change colour, shape and form. they can hide dental wear too, covering abrasion and erosion sites, together with covering over hairline fractures and internal staining.
dental veneers can be made from porcelain or from resin composite materials.
porcelain veneers resist stains better than resin veneers and better mimic the light reflecting properties of natural teeth. they are made in the laboratory.
resin or composite bonded veneers are thinner and require removal of less of the tooth surface before placement. they are made chair side.
minimally invasive, every case requires a different amount of tooth removal, if any. Typically, however, the procedure involves removing 0.3-0.5mm from the outward surface of the tooth. a layer of porcelain or composite (veneer) is then placed onto the surface in the colour, shape and position required.
pros
- they provide a natural tooth appearance.
- gum tissue tolerates porcelain well.
- porcelain veneers are stain resistant.
- the colour of a porcelain veneer can be selected such that it makes dark teeth appear whiter.
- veneers offer a conservative approach to changing a tooth’s colour and shape.
- tooth veneers generally don’t require the extensive shaping prior to placement that crowns do, yet offer a strong and aesthetic alternative.
cons
- the process is irreversible.
- veneers are more costly than composite resin bonding.
- tooth veneers are usually not repairable should they chip or crack.
- because enamel has been removed, your tooth may become more sensitive to hot and cold foods and beverages.
- veneers may not exactly match the color of your other teeth. also, the veneer’s color cannot be altered once in place. if you plan on whitening your teeth,
- you need to do so before having veneers cemented.
- though not likely, veneers can dislodge and fall off. to minimize the chance of this, do not bite your nails, chew on pencils, ice, or other hard objects.
- teeth with veneers can still decay, possibly necessitating full coverage of the tooth with a crown.
- veneers are not a good choice for individuals with unhealthy teeth (for example, those with decay or active gum disease), weakened teeth (as a result of decay, fracture, large dental fillings), or for those who have an inadequate amount of existing enamel on the tooth surface.
- individuals who clench and grind their teeth are poor candidates for porcelain veneers, as these activities can cause the veneers to crack or chip.
longevity – tooth veneers are long-term restorations generally lasting between 5-15 years.
tooth contouring and bonding
‘to mould or shape so as to fit a certain configuration’
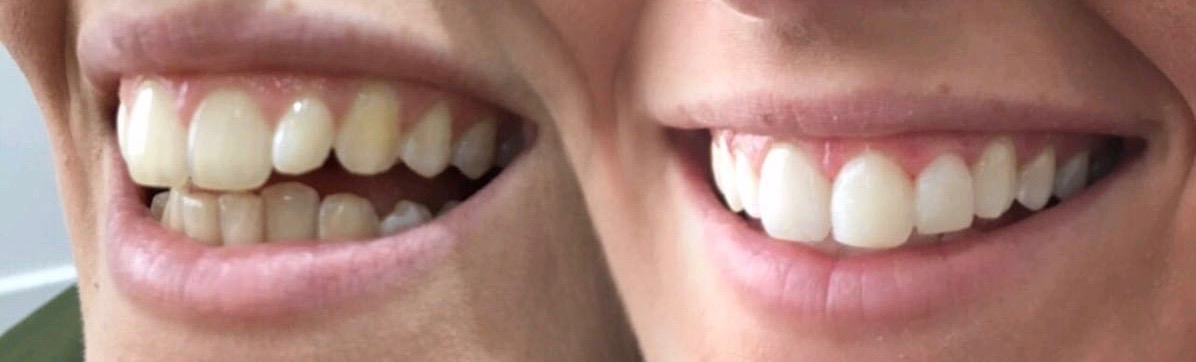
tooth contouring and bonding are cosmetic dentistry procedures that deal with subtle changes to the teeth. often, the slightest change to a corner, or edge of a tooth achieves the desired symmetry and perfects the existing smile-line.
using the rules of ‘the golden proportion’, smiles can be tweaked using a combination of enamel removal (contouring) and sculpted tooth-coloured filling material (bonding). in this way, symmetry is attained and tooth chips along with other imperfections are filled in. polishing discs are also used to flatten rough edges and round off corners.
the technique is minimally invasive and best for people who just need tiny adjustments or repairs to improve the look and feel of their teeth. it can be used to disguise fluorosis, discolouration, round off ‘fangs’, shorten or lengthen teeth. the possibilities are endless, the results astounding.
the pro’s of this type of cosmetic procedure are many. it is relatively inexpensive. because only surface enamel is removed in tooth contouring, there’s generally little pain during, or after the procedure and no anesthesia is required. removing minor overlaps and other imperfections where tartar and plaque can build up lowers your risk of decay and gum disease. in some situations, this conservative approach, can be a substitute for braces as they make teeth look a little straighter and eliminate minor overlapping.
tooth contouring and bonding are quite often, but not always, the first steps toward more extensive cosmetic dental work. procedures such as veneers, tooth whitening, and crown lengthening can be performed months or even years afterward.
minimal, painless, cheaper… but oh-so effective.
facial aesthetics
‘to soften expression lines and define facial contours’
![]() it is often said that aging is a process best done with grace. behind every wrinkle, every crease lies the story of a life well-lived.
it is often said that aging is a process best done with grace. behind every wrinkle, every crease lies the story of a life well-lived.
those stripes have been earned!
and if you don’t mind… it doesn’t matter.
however, if you do mind then there are an array of non-surgical treatments available which, when used correctly, can iron out certain areas.
they say ‘beauty is in the eye of the beholder’ and if what you behold is not to your liking, we are able to help.
the most common of these are:
wrinkle relaxers
a treatment to temporarily improve the appearance of both moderate to severe frown lines between the brows and improve ‘crow’s feet’ in adults. injections can be used to relax certain facial muscles which means that the skin stays smoother and creases gradually diminish.
the result is a more serene, youthful look
dermal fillers
dermal fillers can be used to plump out the facial lines and increase the volume and hydration of the skin. They are used particularly to soften the lines around the mouth and chin area and for enhancing the shape and volume of the lips. These types of fillers are naturally biodegradable and are therefore non-permanent.


